Maduropeptin : Cluster information
 Compound
Compound
| Entry name | Maduropeptin |
|---|---|
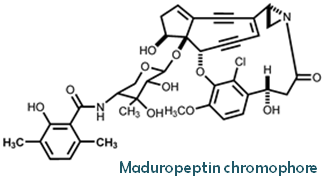
| Moiety description | A: enediyne core B: 3,6-dimethylsalicylic acid moiety C: (S)-3-(2-chloro-3-hydroxy-4-methoxyphenyl)-3-hydroxypropionic acid moiety |
| PKS Type | A: TypeI iterative B: TypeI iterative C: NRPS |
| Starter Unit | A: acetyl-CoA B: acetyl-CoA |
| Chain Length | A: 8 B: 4 |
| Sugar Unit | madurose |
| Classification | Enediyne |
| Activity | Antibiotic Antitumor |
| Composition | C39H39ClN2O11 |
 Original source
Original source
| Organism | Actinomadura madurae |
|---|---|
| Strain | ATCC 39144 |
| Contig | AY271660 |
 PKS/NRPS Module
PKS/NRPS Module
| A | Madu_00440 mdpE | 1 |  | acetyl-CoA malonyl-CoA |
|---|---|---|---|---|
| B | Madu_00170 mdpB | 1 |  | acetyl-CoA malonyl-CoA |
| C | Madu_00290 mdpC5 | 1 |  | |
| Madu_00320 mdpC1 | 1 |  | L-alpha-tyrosine | |
| Madu_00330 mdpC2 | 1 |  |
 Reference
Reference
- A genomics-guided approach for discovering and expressing cryptic metabolic pathways.
- Zazopoulos E, Huang K, Staffa A, Liu W, Bachmann BO, Nonaka K, Ahlert J, Thorson JS, Shen B, Farnet CM[PMID: 12536216]Nat Biotechnol. 21 (2003) 187-90
- Characterization of the maduropeptin biosynthetic gene cluster from Actinomadura madurae ATCC 39144 supporting a unifying paradigm for enediyne biosynthesis.
- Van Lanen SG, Oh TJ, Liu W, Wendt-Pienkowski E, Shen B[PMID: 17918933]J Am Chem Soc. 129 (2007) 13082-94
- Enediyne antitumor antibiotic maduropeptin biosynthesis featuring a C-methyltransferase that acts on a CoA-tethered aromatic substrate.
- Ling J, Horsman GP, Huang SX, Luo Y, Lin S, Shen B[PMID: 20718468]J Am Chem Soc. 132 (2010) 12534-6
- Enediyne polyketide synthases stereoselectively reduce the beta-ketoacyl intermediates to beta-D-hydroxyacyl intermediates in enediyne core biosynthesis.
- Ge HM, Huang T, Rudolf JD, Lohman JR, Huang SX, Guo X, Shen B[PMID: 25019332]Org Lett. 16 (2014) 3958-61
 Data download
Data download
 History
History
- 2016-01-27[Update]
- 2015-02-12[Update]
- 2013-12-27[Release]